Regenerative medicine

What is regenerative medicine?
Practical applications of nerve regeneration
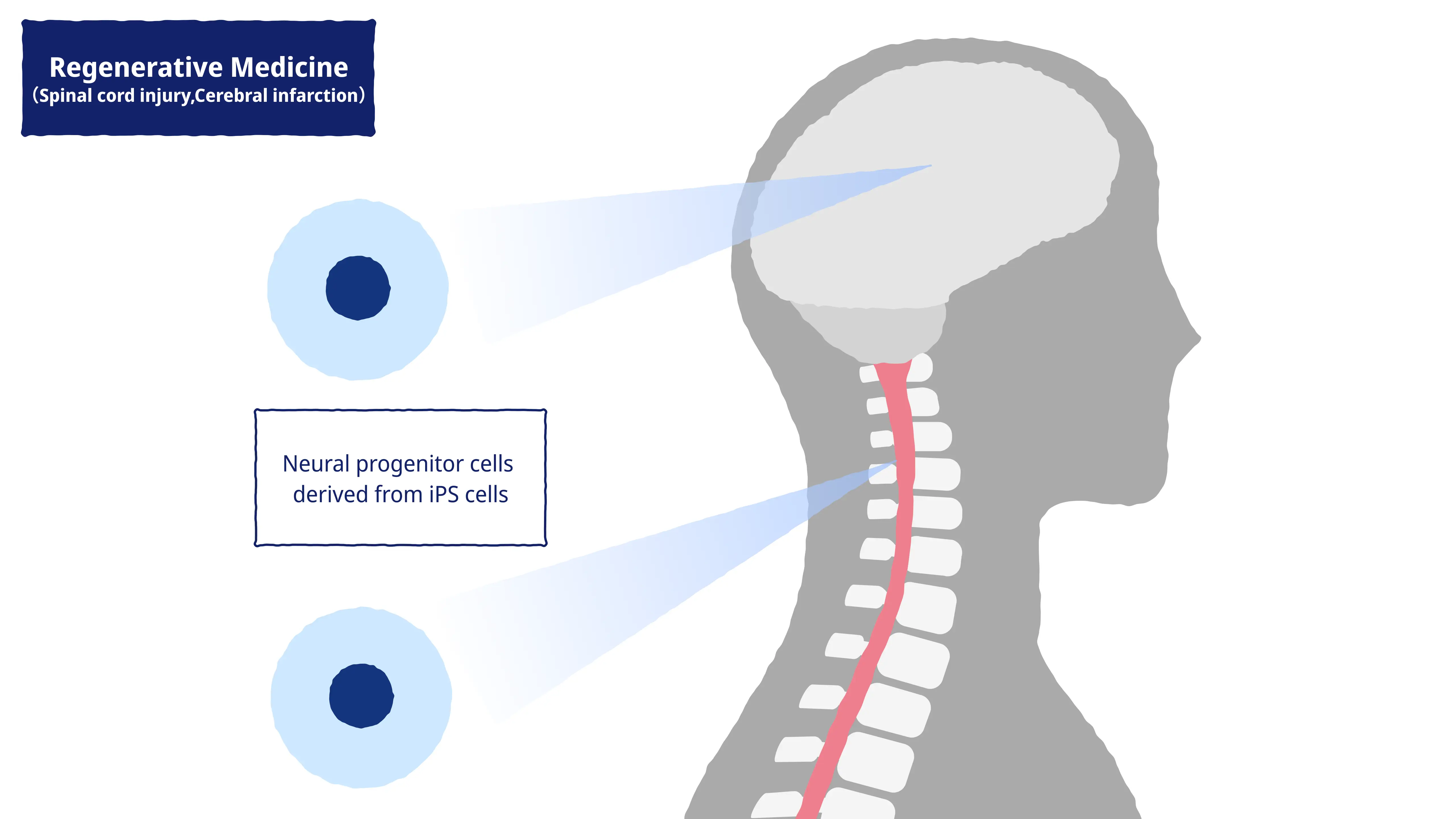
Regenerative medicine is the science of using iPS cells to regenerate tissue and internal organs lost through disease or mishap in order to restore or enhance functioning. There is strong demand at the global level to develop therapeutic treatments for patients with unmet medical needs, and regenerative medicine represents an exciting new approach with considerable potential in this field.
K Pharma is dedicated to the study of iPS cells in regenerative medicine to treat neurological conditions such as cerebral infarction and SCI. Although regeneration as a technique has traditionally been considered unsuitable in the neurological domain, practical applications for nerve regeneration are now closer than ever, thanks to cutting-edge research performed by professors Hideyuki Okano and Masaya Nakamura at the Keio University School of Medicine over many years, combined with the independent resources of K Pharma today.
Main disease areas
Learn about the diseases we are tackling through regenerative medicine.
-
Subacute spinal cord injury
Spinal cord injuries (SCI) are caused by compression of the spinal cord, typically due to a dislocation or fracture event. The term subacute SCI refers to an injury that has stabilized and is no longer in the acute phase. This is when angiogenesis and the tissue repair response are at their strongest, which can potentially lead to paralysis and other full-body complications.
Chronic spinal cord injury
A chronic spinal cord injury is one that persists well after the initial damage to the spinal cord. It is caused by the formation of very hard tissue at the injury site, which makes it harder to improve motor function through conventional therapeutic treatment.
-
Cerebral infarction
Cerebral infarction occurs when blood flow to any part of the brain is restricted, which can occur for a number of different reasons. This leads to impaired brain functioning that can manifest in a variety of ways, such as numbness or paralysis of limbs on one side, difficulties with speech, and consciousness disorder.
Business environment
Learn about the latest developments in the domestic and global pharmaceutical industry.
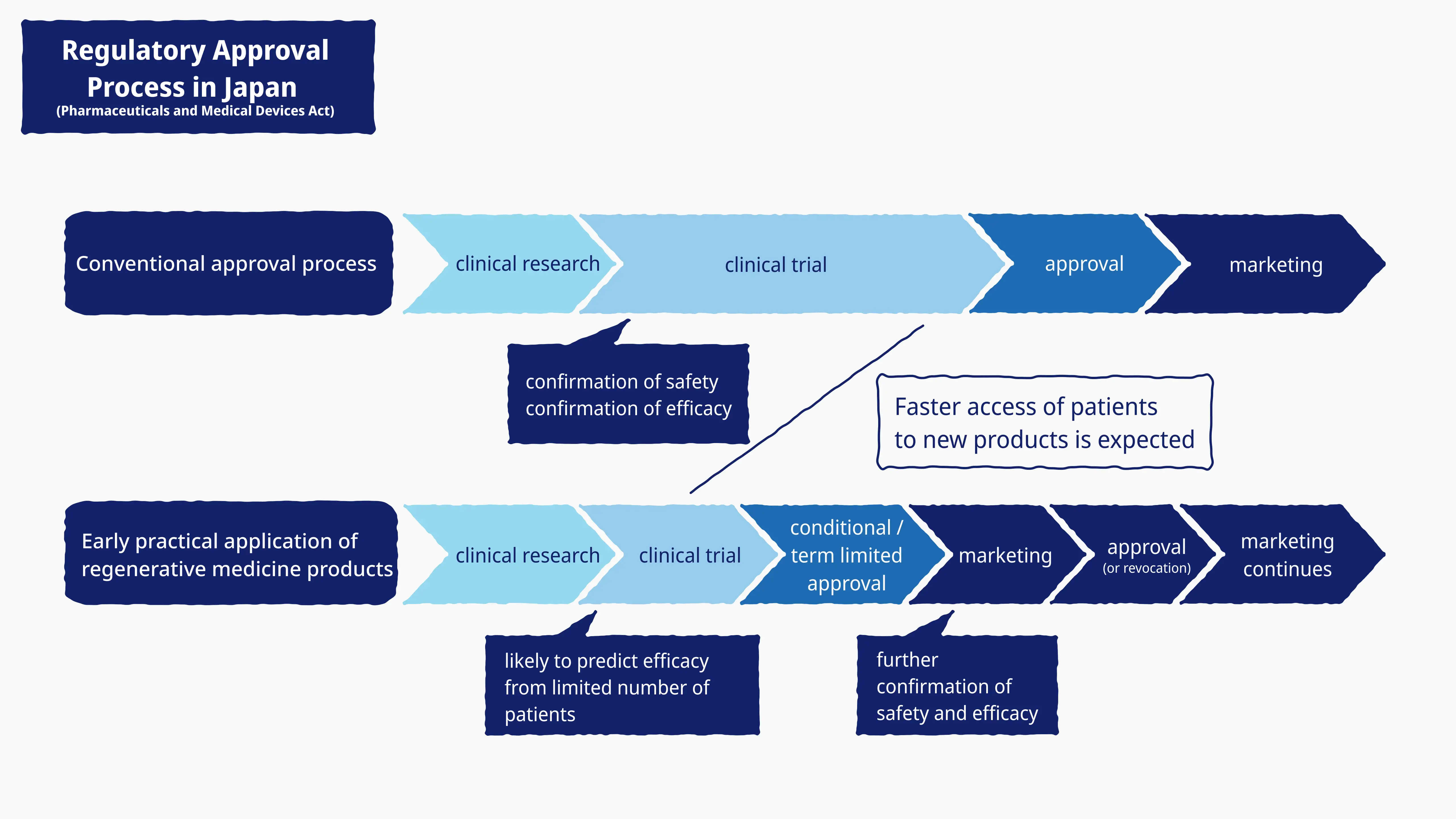
Fast-track approvals bring regenerative medicine a step closer
The Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices, enacted in 2014 through amendments to the previous Pharmaceutical Affairs Law, defines “regenerative medicine products” separately to pharmaceuticals and medical devices, and allows for “fast-track” approvals in cases where efficacy is presumed and safety has been demonstrated. Fast-track approvals are time-limited and subject to restrictive conditions. This important legislative change has provided added impetus to our mission to develop practical applications for nerve regeneration technology as quickly as possible.
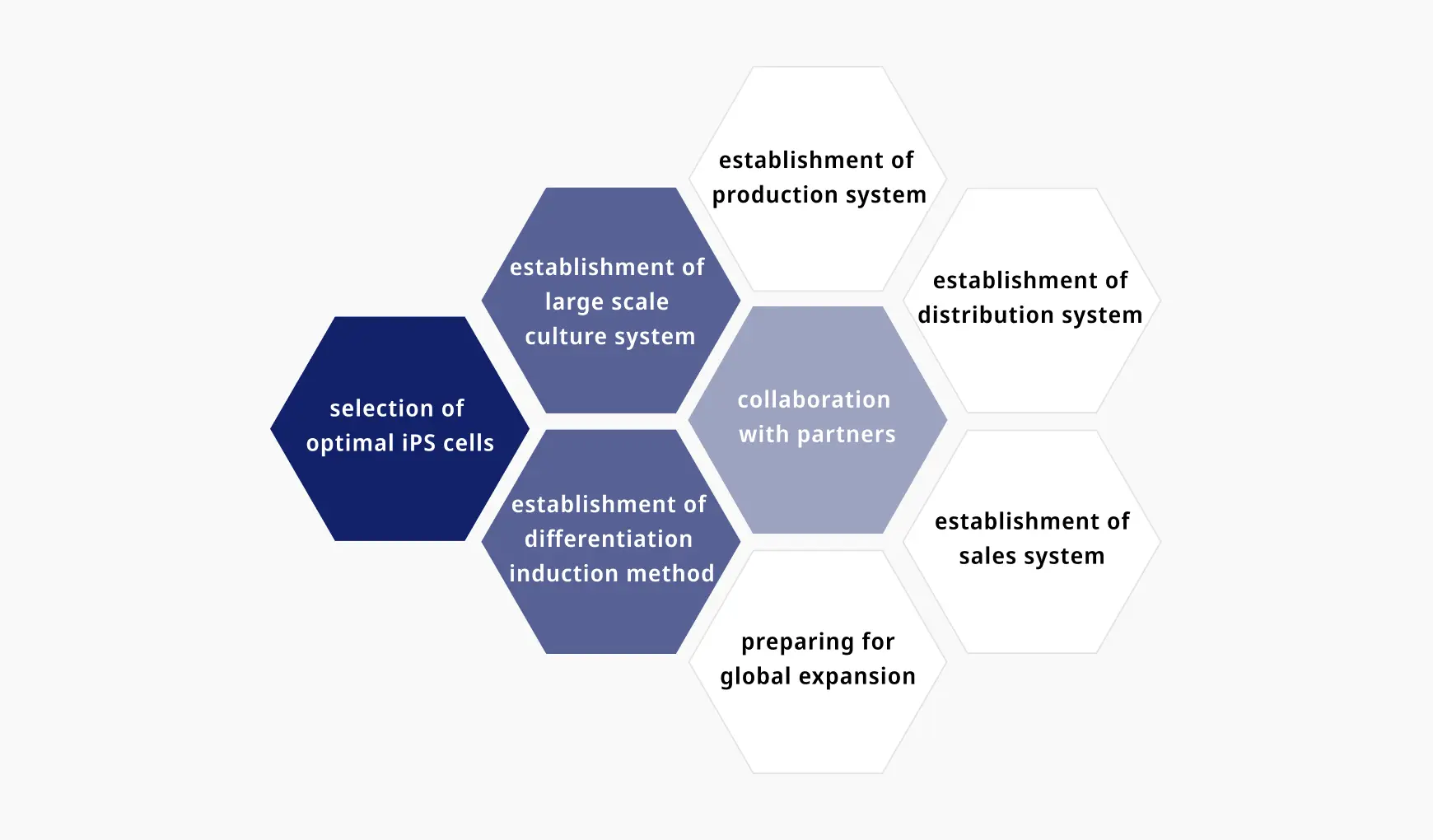
Development focus
From Basic to Clinical
K Pharma is dedicated to using our world-leading research to develop regenerative medicine treatments for spinal cord injuries and intractable diseases, with a view to a global roll-out in the future.
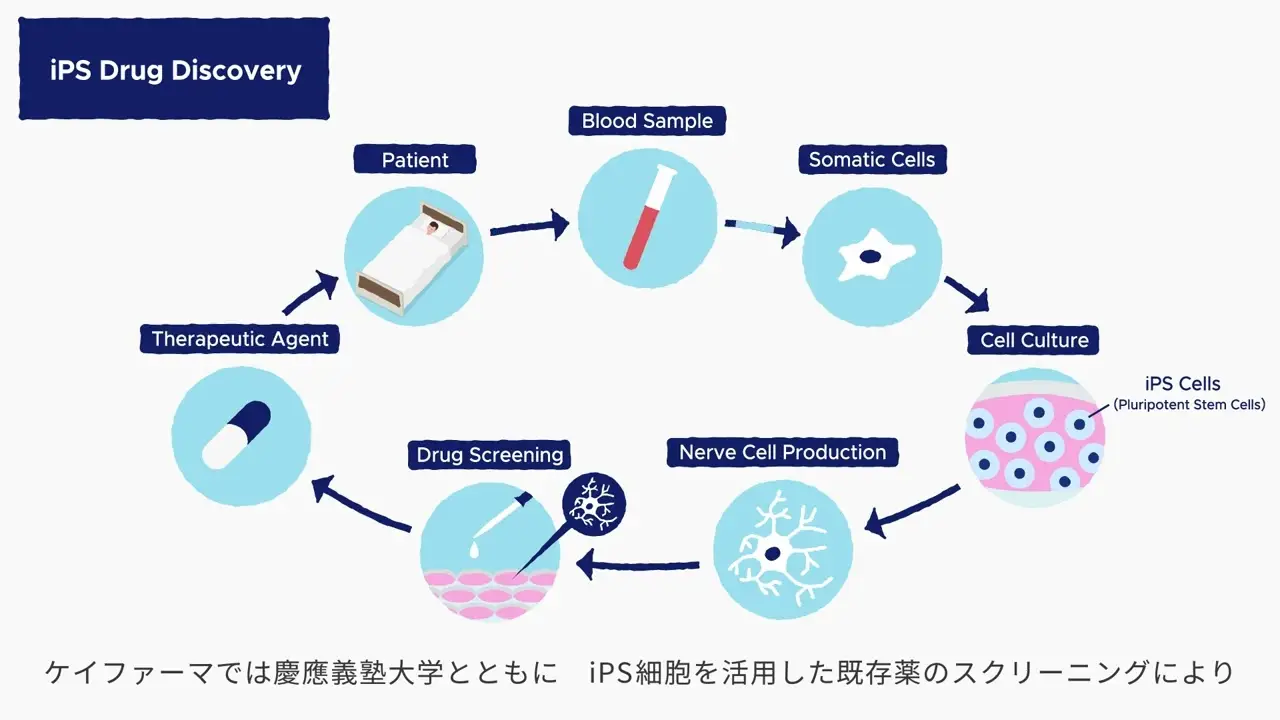
This video describes our work in iPS drug discovery and regenerative medicine.